Stem cells are highly desirable cells because of their elasticity and ability to be turned into a wide variety of cell types. These cells are most often thought of for their regenerative properties and application for therapies. However, stem cells have a diverse array of applications, one of which includes the production of artificial, lab grown meat.
What is artificial meat?
Artificial meat, otherwise known as cultivated meat or cultured meat, is genuine animal meat that is produced by culturing animal cells. In this process, the animal’s stem cells are collected and grown in a lab, creating meat products without killing the animal. This method provides an alternative way of sourcing animal-based foods.
To make artificial meat, the collected stem cells are grown in large-scale bioreactors. In these chambers, cells are provided the proper nutrients and growth factors needed to maintain, and ultimately scale, stem cell growth. From there, the stem cells are differentiated into muscle, fat, and connective tissue cells needed to create meat. This differentiation is done by feeding the stem cells the appropriate nutrients and growth factors needed to signal their transition into the new cell type.
Is artificial meat better?
The subjectivity of taste and flavor aside, consumers still have mixed thoughts about this new type of meat. Many people fear that meat grown from a dish is unclean, unethical and will be very expensive. However, artificial meat could have many economic benefits.
Since artificial meat does not require the need to maintain livestock, it will use less resources and reduce agricultural pollution. Cattle farms consume large amounts of natural resources, to the point where they have been criticized for their contribution to deforestation. Unlike many livestock, artificial meat can be grown without antibiotics or hormones and has less risk of contamination.
That said, artificial meat still faces criticism because raw materials needed to grow and maintain these cells in culture includes fetal bovine serum, a serum sourced from slaughtering pregnant cows. This material is very expensive and still results in animal cruelty. However, companies are working to source ethical alternatives to these materials, creating what might be the cleanest meat to exist.
Will cultured meat ever replace natural meat?
It is hard to say if artificial meat will ever be our sole source of animal-derived proteins, but companies are racing to be the first to bring artificial meat to the market. As of 2022, it was reported that 150 companies globally are dedicated to artificial meat production – all supported by over $2.6 billion and up from only 4 companies in 2015. Two companies, Mosa Meat and Aleph Farms, even have Leonardo DiCaprio’s financial backing and support! However, in the United States, federal approval from the Food and Drug Administration (FDA) have delayed cultivated meat products from reaching the market.
Currently, cultured meat sells at higher prices per pound compared to their traditional counterparts which has been a barrier to entry. As more approvals are granted and demand increases, manufacturing artificial meat would increase in scale, ultimately allowing cultured meat prices to match or even best current meat prices. With this scale up, however, comes another set of challenges companies must be ready to tackle. There is a lot of work to be done before we are all being served lab grown meat, but it might be an option sooner than we might think.
How can we make artificial meat production more efficient?
One of the critical aspects of artificial meat production is the process of growing and maintaining high quality stem cells. The higher the quality of the stem cells, the easier it is to differentiate them into meat-specific cell types. This also allows for more efficient production which in turn reduces the cost.
StemCultures specializes in the manufacture of controlled-release growth factor technology to optimize cell culture conditions. Growth factors needed to maintain cells have very short half-lives. One of the most critical growth factors for maintaining stem cells is FGF2, which has a half-life of 4.5 hours. Because of this, growth factor levels fluctuate drastically from day to day, creating a stressful environment for cell growth.
Is controlled-release technology the key to combating artificial meat criticisms?
By maintaining a stable level of the growth factor in culture, cells are guaranteed to always be supplied with the nutrients critical for survival and maintenance. This creates a favorable environment for cell growth, leading to the highest quality results. It might also prove to eliminate the need for unethically sourced animal-based serums. Not to mention, all of this is done with a reduction in feeding schedules. Instead of daily cell maintenance, scientists only have to perform medium changes 2-3x per week, further making the process more efficient and cost effective.
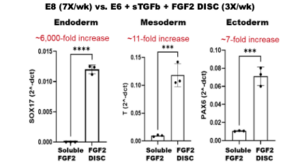
Stem cells grown using StemCultures’ FGF2 DISCs show significantly more efficient differentiations into each of the three cell type lineages (figure below). Across different PSC media backgrounds, we observed improved directed differentiation efficiency when the stem cells were cultured with FGF2 DISCs (and reduced feeding) prior to differentiation.
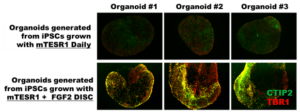
Organoids generated from stem cells grown with FGF2 DISCs and reduced feeding patterned better than those grown with mTeSR1 and daily medium changes (Figure below). This goes to show that organoids, and other down stream applications of stem cells, require high quality cells to have robust outcomes.
Applying controlled-release growth factor technology to stem cells grown for artificial meat production could overcome manufacturing inefficiencies, reducing costs, improving cell quality, and giving an option to replace animal-derived serums.
—
Note: Opinions and accounts expressed herein are those of the author(s) or interviewee(s). They may not reflect those of StemCultures, its officers, or directors.